Why Use Pentane, a Hydrocarbon?
Polyisocyanurate foams were traditionally produced using CFC-11 (a chloro-fluorocarbon) as the blowing agent. When evidence became irrefutable that CFCs destroyed stratospheric ozone, most of the world adopted the ground-breaking Montreal Protocol, which mandated the phaseout of CFCs for non-essential uses by 1996. Many polyiso producers gradually transitioned to HCFC-141b (a hydro-chlorofluorocarbon), which has only 10% to 12% the ozone-depletion potential of CFC-11. But since HCFC-141b was recognized as the most damaging of the HCFCs, HCFC-141b would be only a temporary solution. (Modifications to the Montreal Protocol later mandated the phaseout of this chemical by 2003.)
As polyiso manufacturers studied possible substitutes for HCFC-141b, two different hydrofluorocarbons emerged as possible substitutes: HFC-245FA and HFC-365. HFCs have the advantage of being non-ozone-depleting (since they don’t contain chlorine or bromine), but they are significant greenhouse gases. Most HFCs are also expensive to manufacture.
Another alternative was a hydrocarbon blowing agent – – pentane. Hydrocarbon blowing agents have the advantage of being less expensive, but their flammability requires special safety measures at manufacturing plants. Yet the cured foam is no more flammable than HCFC-blown foam.
As for energy performance, leading industry experts report there is no appreciable change in R-value with the hydrocarbon-blown foams. The finer cell structure of pentane-blown foams, for instance, tends to offset the pentane’s higher thermal conductivity. Pentane-blown foams have another advantage: better dimensional stability due to the fact that pentane does not condense as much as HCFC-141b at temperatures normally experienced by the foam in use. The condensation of HCFC-141b causes the cells to shrink and expand on a cyclical basis, reducing dimensional stability.”
 An Insulation System For Each Temperature Range
An Insulation System For Each Temperature Range
Rigid Foam Insulation and the Environment
Ozone depletion and global warming are two of our most serious environmental problems—and foam insulation materials containing CFCs (chlorofluorocarbons) contribute significantly to both of these problems. The environmentally concerned builder or designer should make it a highest priority to avoid them. Even many of the non-CFC alternatives that manufacturers are now switching to are still damaging to the environment—though less so than CFCs. This article takes a detailed look at various types of foam insulation materials—how the materials are produced, what their environmental impacts are, and what the alternatives available to you are.
Rigid foam has played an important role in the energy-efficient construction revolution we have witnessed since the mid-’70s, permitting wall and roof R-values to be boosted dramatically with only minimal increases in wall thickness. The foam has such a significant effect in part because it covers the framing members, thus reducing the thermal bridging that occurs through framing members when only cavity-fill insulation is used. The increasing popularity of foam-core stress-skin panels—some of which are produced with CFC-based foams—for use in both timber frame and structural panel (frameless) buildings is further increasing use of environmentally damaging foams.
Types of Rigid Foam Insulation
There are several major types of rigid foam insulation, including polyisocyanurate (which is a type of polyurethane), extruded polystyrene, phenolic foam, and expanded polystyrene (EPS). The first three are described below, with information on their general properties and how they are produced. EPS, which does not cause ozone depletion, is discussed later.
Polyisocyanurate
Polyisocyanurate insulation (iso) is widely used in the construction industry, with some 2.5 billion board feet produced in North America in 1989, according to the Society of the Plastics Industry. Iso insulation is typically foil-faced, and it is widely available in thicknesses from
1/2” to 4″. Common trade names for iso insulation include R-max, Thermax, Tuff-R, Energy Shield, ACFoam, and ENRGy 1. It is also used in certain Exterior Insulation and Finish Systems (EIFS), and in foam-core stress-skin panels made by Winter Panel Corp., Atlas Industries, and several other manufacturers.
CFC-11 is used in the production of iso to generate rapid expansion of foam with a closed-cell structure and to provide high R-values—about R-7 per inch. (This R-value drops slowly over time as air leaks into the cells and as CFC-11 leaks out.) Most iso foams are 11 to 15 percent CFC-11 by weight. In the past two years, manufacturers have succeeded in reducing the amount of CFC-11 in these foams by improving manufacturing efficiencies, reducing waste, and adding water to the mixture, which generates CO2 during the foaming process. Throughout the polyisocyanurate insulation industry, the CFC-11 content has dropped by as much as a third—from 15% to 10-12%, according to Jared Blum of the Polyisocyanurate Insulation Manufacturers Association.
While short-term efforts to reduce CFC-11 use by improving efficiency and adding water to the chemical mixture have been moderately successful, most efforts are focusing on the intermediate future—from 1994 through 2015. In this time-period iso manufacturers are expected to switch to hydrochlorofluorocarbon (HCFC) foaming agents. The industry has identified HCFC-141b as the most promising replacement for CFC-11. (Another likely candidate, HCFC 123, was dropped when early toxicity testing turned up tumor formation in laboratory rats.) Results of performance testing of HCFC-141b-based iso foams have been quite positive. Although the resulting R-value is 4% to 8% lower than that of foam produced using CFC-11, the drop in R-value over time appears to be slower.
Extruded Polystyrene
Extruded polystyrene was invented in Sweden, but the product was further developed in the United States during the 1940s. It is produced by four manufacturers in North America, all of which claim an insulating value of R-5 per inch. Extruded polystyrene has become the insulation of choice for most below-grade applications, and it is widely used for wall sheathing as well.
Originally, methyl chloride was used as the blowing agent, but manufacturers switched to CFC-12 during the 1960s. CFC-12 was less toxic, nonflammable, and provided a higher R-value. The foam is approximately 10 percent CFC-12 by weight at the time of manufacturing, with roughly 85 percent retained after production. In 1986, use of CFC-12 for extruded polystyrene totaled approximately 39 million tons worldwide.
By the late 1980s, with increasing pressure to eliminate CFC use, most extruded polystyrene manufacturers began switching to a mixture of HCFC-142b and ethyl chloride as the foaming agent. HCFC-142b is approximately 15 percent more expensive than CFC-12, thus raising the cost (or reducing the profit margin) of foam produced with it, but its ozone depletion potential is 94% less than that of CFC-12.
Amoco Foam Products fully converted to the HCFC-142b/ethyl chloride mixture in its Amofoam® product line by January 1990. Dow Chemical completed the conversion with its Styrofoam® product line by July 1990 (with a slightly different mixture from Amoco’s). The two other manufacturers, UC Industries and Diversifoam, continue to use CFC-12. UC Industries, maker of FoamulaR™, had converted approximately 50 percent of its production away from CFC-12 by mid-1992 and intends to complete the conversion by the end of the year. Diversifoam Products, maker of Certifoam®, is also in the process of conversion but did not provide a timetable for phasing out CFC-12. In addition to leading the way in eliminating CFC-12, Amoco Foam Products has also just introduced the Amofoam-RCY product, made with 50 percent recycled polystyrene.
Phenolic Foam
Even though it was quite popular with some energy-conscious builders because of its very high R-value, phenolic foam insulation is no longer in production in the United States. Manville Corporation, which purchased the phenolic foam production rights from Koppers a few years ago, suspended production of its Ultra Guard Premier in February of this year. Bert Emory of the company cited a limited customer base and lack of profitability for the decision to cease production, but another source indicated that the reason had to do with the cost of replacing the CFCs.
Two companies in Canada still produce phenolic foam insulation: Domtar and Fiberglas Canada. With no iso manufacturers in Canada, phenolic foam has a sizeable share of the Canadian boardstock insulation market.
The Fiberglas Canada product, Perma-Therm, is sold only for commercial roofing applications at this time, though the company is considering expanding into residential roofing and wall applications. Their product, which is actually a “modified resol” product (resol is a type of phenol resin), was made until 1990 using CFC-114 as the blowing agent. Jim Sidwell, Fiberglas Canada’s Business Manager for Commercial Roofing Products, said that the company has already reduced its CFC-114 use by 80% and is now producing the foam insulation using a 20:80 mix of CFC-114 and an unspecified HCFC. Legislation in Ontario calls for a 50% reduction in CFC use by 1992, a 75% reduction by 1993, and a 100% reduction by 1994. Sidwell expects the company to complete its switch to HCFCs by the first half of 1993.
Domtar produces a phenolic foam insulation board under the Rx brand name (the same brand name Koppers used, since identical technologies were licensed to the two companies). Domtar sells about 50 million board feet of the product per year for both wall and roof applications. Domtar’s Rx phenolic board is produced using a 50:50 mixture of CFC-11 and CFC-113. Because of the manufacturing process, they have been able to use recycled CFC-113, even with some impurities. (By using recycled foaming agents, they satisfy the Ontario requirement for 50% reduction in CFC use this year.) Domtar expects to have eliminated use of CFCs totally by the end of the year.
Just How Bad are CFCs for the Environment, Anyway?
Pretty bad. The realization that CFCs could deplete the Earth’s stratospheric ozone layer has led to unprecedented international cooperation in banning these compounds. It was first suggested in 1974 that CFCs could destroy ozone in the stratosphere. CFCs are highly stable chlorine-containing compounds that do not break down under normal exposure to sunlight and moisture as most organic compounds do. According to current understanding, once released into the atmosphere they gradually make their way up to the stratosphere where high-energy ultraviolet light can break the molecules apart, releasing chlorine atoms. The chlorine atoms then react with ozone (O3) by bonding with one oxygen atom to form chlorine monoxide. The chlorine monoxide itself is unstable and quickly breaks down by reacting with another ozone molecule. A single chlorine atom can destroy as many as 100,000 ozone molecules.
After a dozen years spent challenging the underlying theories and making only minor progress toward eliminating the harmful compounds, international interest was piqued in 1985 with the discovery of a hole in the ozone layer over Antarctica. This led to passage of the Montreal Protocol in 1987 calling for a 50% reduction in CFC production by 1998. Following the discovery that ozone thinning was taking place over the far-more-populated Northern Hemisphere, the Montreal Protocol was strengthened in 1990. The so-called London Amendments call for a total phaseout of CFCs and halons (fire-extinguishing agents) by the year 2000 and phaseout as well for carbon tetrachloride and methyl chloroform. By 1992, more than 70 countries, representing 90% of the world’s CFC production, had signed onto the strengthened Montreal Protocol.
Such rapid and decisive international action has been taken because of the serious threat posed by ozone depletion. Stratospheric ozone protects us by blocking out high-energy ultraviolet radiation (UVb), which has harmful health and environmental effects. According to a report compiled for the U.S. Environmental Protection Agency, the extra UVb radiation reaching the Earth’s surface because of ozone depletion will cause over 900,000 cases of nonmelanoma and melanoma skin cancer in the United States (including 14,600 fatalities) and 160,000 cases of cataracts among people born prior to 1986—even with the CFC phaseout treaty currently in place. Had CFC controls not gone into effect, total cases of skin cancer would exceed 6 million, including more than 120,000 fatalities, and over 1.8 million cases of cataracts among U.S. citizens born prior to 1986. In addition, increased UVb radiation would be expected to harm marine organisms (including commercial fisheries), agricultural crops, and polymers exposed to sunlight. The report doesn’t attempt to evaluate damage to natural ecosystems that do not have recognized commercial value, but the harm to all ecosystems would likely be significant.
 Table 1.
Table 1.
Environmental Impact of Foaming Agents Used in Rigid Insulation
Sources:
1. D. Fisher, et al., “Model calculations of the relative effects of CFCs and their replacements on stratospheric ozone,” Nature, Vol. 344, 5 April 1990. One-dimensional model numbers from DuPont.
2. Regulatory Impact Analysis: Compliance with Section 604 of the Clean Air Act for the Phaseout of Ozone Depleting Chemicals, Prepared for the U.S. Environmental Protection Agency by ICF, Inc., March 12, 1992. Global Warming Potentials based on infinite time horizon.
Not all foaming agents are equally destructive of ozone. Some CFCs last longer than others in the atmosphere, some contain more chlorine, and HCFCs contain hydrogen in addition to chlorine, making them less stable and more likely to break down before reaching the stratosphere. “Ozone depletion potential” (ODP) is a measure of the relative potency of different chemicals in terms of ozone destruction. ODPs are generally measured relative to CFC-11, which is defined to have an ODP of 1.0. Ozone depletion potentials of various foaming agents are shown in Table 1. Note that the alternatives to CFCs for foam insulation materials (HCFC-141b and HCFC-142b) have considerably lower ODPs than the CFCs, but even these compounds have a sizeable effect on ozone.
One could argue that CFCs from foam insulation materials are not that much of a concern since the CFC gas is locked up in the foam. Indeed, some measurements show that CFC-11 in iso foam has a half-life of more than 100 years. (The drop in R-value over time observed with CFC-blown insulation materials, according to an expert at DuPont Corporation, is more a result of air leaking into the cell structure than CFC-11 leaking out.) The long lifetime of CFCs in foam insulation, however, does not absolve them from environmental concern. In fact, one could argue equally well that the long lifetime of CFCs makes them worse from an environmental standpoint, because even when all CFC production halts, these foams will continue releasing CFCs into the atmosphere for hundreds of years.
Along with depleting ozone, CFCs are potent greenhouse gases that are implicated in global warming. Carbon dioxide (CO2) is the most significant contributor to global warming because of the enormous amounts generated. Pound for pound, however, CFCs are far more significant contributors to global warming. One pound of CFC-11 is equivalent to 1600 pounds of CO2 in terms of global warming potential, and a pound of CFC-12 is equivalent to 4400 pounds of CO2. While the HCFCs are not as bad as CFCs, they are still far worse than CO2. In fact, HCFC-142b, which is being used in extruded polystyrene, is nearly a third as detrimental as CFC-11 from a global warming standpoint.
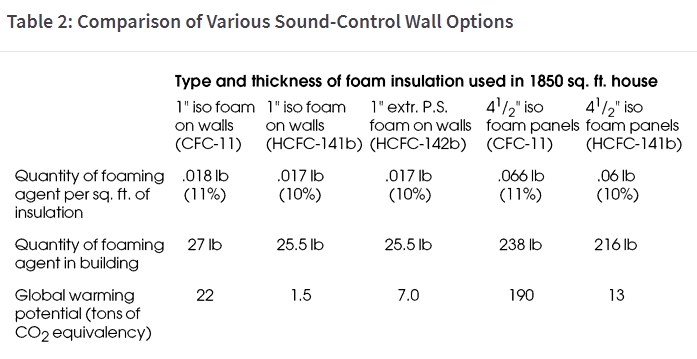
Table 2 shows the relative global warming impact of houses with different wall and roof insulation systems, relative to comparable CO2 emissions. The results are striking and frightening. An average-size house with 1” of iso foam on the walls will introduce 27 pounds of CFC-11 into the atmosphere over time, which is comparable to 22 tons of CO2 emissions. A foam-core panel house, with 41/2” isocyanurate-core panels, contains 238 pounds of CFC-11 with as much global warming impact as 190 tons of CO2 emissions! If that house uses 500 therms of natural gas per year for heating, it would take 63 years for the CO2 emissions from the natural gas combustion to equal the global warming potential in the foam.
The widespread use of foam in commercial roofing is even more troubling, since common roofing practices scrap the entire mass of insulation each time the roofing surface is replaced. This insulation, then, has a useful life of only about twenty years, after which it becomes a part of the landfill problem, while continuing to release CFCs into the atmosphere.
HCFC foams are better relative to global warming than CFC-based foams, but their impact is still significant.
What are the Alternatives?
If we accept that rigid foam insulation materials produced with CFCs and HCFCs are not acceptable from an environmental standpoint, what are the alternatives? Currently, the only non-CFC and non-HCFC boardstock insulation materials on the market are rigid fiberglass and expanded polystyrene (commonly called EPS or beadboard). EPS is produced using pentane as the foaming agent.
From an environmental standpoint, rigid fiberglass—produced from glass fibers held together with a binder—is probably the best boardstock insulation material (though some embodied energy studies indicate that EPS may be superior—this issue will be addressed in a later issue of EBN). Unfortunately, rigid fiberglass is not marketed widely for residential construction in the U.S., though it is available in Canada (GlasClad, produced by Fiberglas Canada, 4100 Younge St., Willowdale, Ontario M2P-2B6). In the U.S., rigid fiberglass is produced for commercial applications by all three major fiberglass manufacturers: Owens Corning, Manville, and Certainteed. Some companies have also worked out cladding systems allowing rigid fiberglass to be used below grade as foundation insulation.
Expanded polystyrene (EPS) is the only foam boardstock insulation made entirely without CFCs or HCFCs. To manufacture EPS, pentane-filled polystyrene beads are expanded using heat, releasing most of the pentane into the atmosphere. A hydrocarbon, pentane emissions contribute to localized air pollution (smog), but its impact on global warming is negligible due to its short lifetime in the atmosphere, and it has no effect on stratospheric ozone.
EPS has long been considered a lower quality product than extruded polystyrene. Its perceived drawbacks relative to extruded polystyrene include lower R-value, inferior structural properties, and possible disintegration over time in below-grade applications. While some low-cost 1 lb/ft3 EPS may indeed have these drawbacks, EPS is also available in higher densities. At 2 lb/ft3, EPS is much closer to extruded polystyrene in its performance and considered an acceptable substitute in both above- and below-grade applications. EPS boardstock is generally available directly from the manufacturers, of which there are several hundred in North America, and some building supply yards may stock it. Because EPS pricing is generally proportional to its density, 2 lb/ft3 EPS will be close to extruded polystyrene in cost.
The other alternative to CFC and HCFC foam insulation materials is to eliminate rigid boardstock insulation from construction details altogether. Fiber insulation materials, including fiberglass, mineral wool and cellulose, are all free of CFCs and HCFCs. To achieve comparable R-values with these materials, you have to build thicker walls, but the higher framing costs should be largely offset by eliminating the expensive rigid foam and the labor required to install it. [See page 8 for one example of a high-R-value wall detail using cellulose.] For heated basements, interior foundation insulation using a studwall with batt or blown-in insulation is better thermally than exterior insulation, and conversion into living space will be much easier.
Foam insulation provides a good example of how a well-meaning push toward energy conservation has led to other problems. Clearly, energy use for heating and cooling buildings is a major cause of pollution, and efforts to reduce that energy use are necessary— indeed a high priority. As new discoveries are made about the effects of our actions on the global environment, however, we have to be willing to adapt our practices in response. Right now, the appropriate response is to develop ways of building that don’t use CFC- or HCFC-based insulation materials. Our challenge is to do so without sacrificing energy efficiency.
– Alex Wilson
Pentane Blends, CAS:109-66-0, in Isotank,15MT/Isotank
pentaneblend #pentaneblends #mixture_of_Isopentane_and Normal_Pentane #CAS109660
n-Pentane, CAS:109-66-0, in 130kg drums and Isotank,160 drums/40″GP, 80 drums/20″ GP, 15MT/Isotank
pentane #normal_pentane #normalpentane #CAS109660 #pentaneblends #pentanes #C5H12 #nPentane Please contact us to request a quote